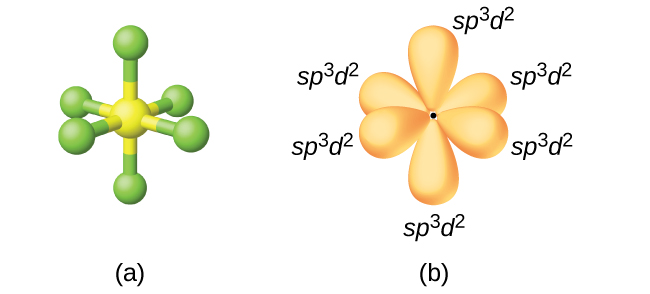
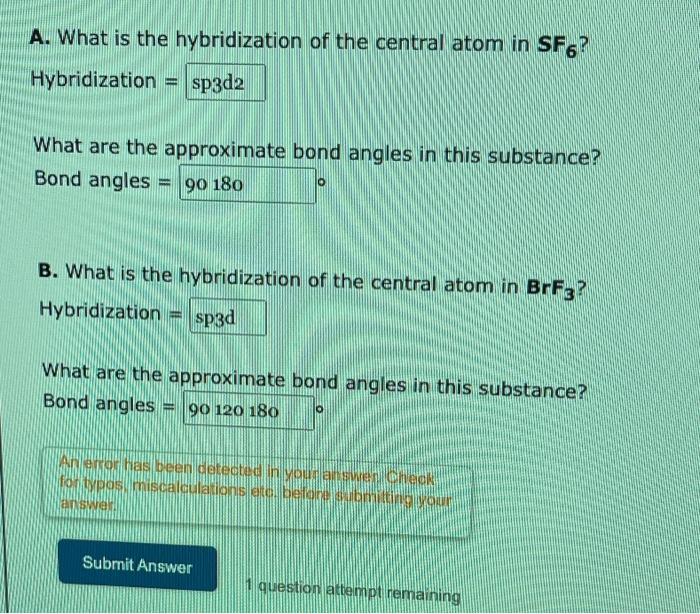
The sp3d2 Hybridization and Octahedral Geometry | Molecular geometry, Chemistry, Electron configuration

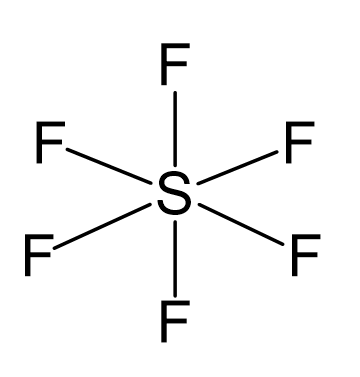
Write the electronic configuration and hybridization of SF6 molecule - Chemistry - - 15171799 | Meritnation.com

Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

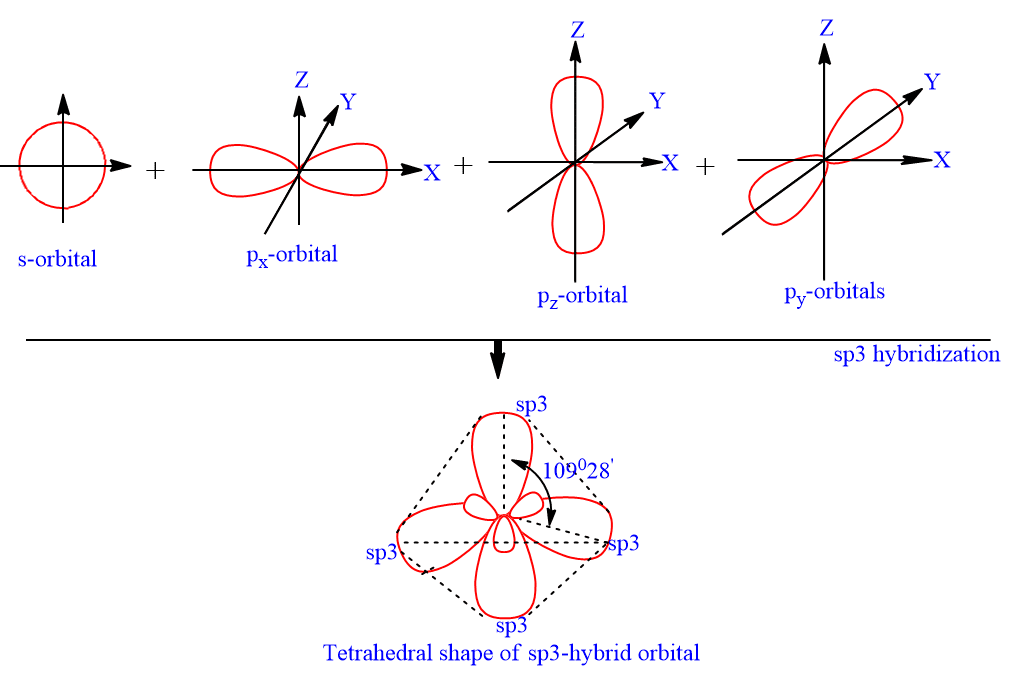
Atomic orbitals involved in hybridization of SF_{6} molecule:3s,3p_{x},3p_{y},3p_{z},3d_{{z}^{2}},3d_{{x}^{2}-{y}^{2}}3s,3p_{x},3p_{y},3p_{z},3d_{{z}^{2}}4s,4p_{x},4p_{y},4p_{z},3d_{{x}^{2}-{y}^{2}},d_{xy}3s,3p_{x},3p_{y},3d_{xy},3d_{yz},3d_{xz}

What atomic or hybrid orbitals make up the sigma bond between S and F in sulfur hexafluoride, SF6? | Homework.Study.com

Describe hybridisation in the case of PCl5 and SF6. The axial bonds are longer as compared to equatorial bonds in PCl5 whereas in SF6 axial bonds and equatorial bonds have the same

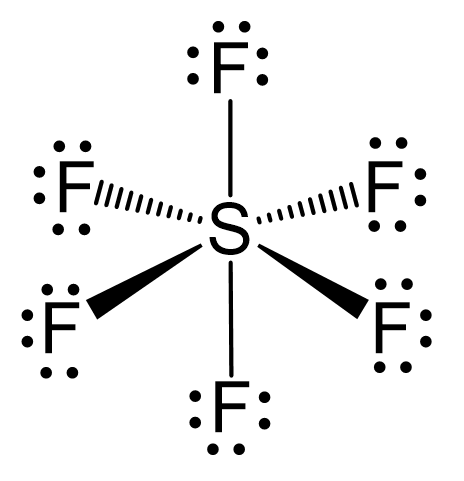
![Solved] In SF6 the hybridisation of sulphur is: Solved] In SF6 the hybridisation of sulphur is:](https://storage.googleapis.com/tb-img/production/21/01/F1_Utkarsha_15.1.21_Pallavi_D27.png)